Non-conformance
(Alias: non-compliance)
Why, in defiance of every precept and principle of this house, does she conform to the world so openly--here in an evangelical, charitable establishment--as to wear her hair one mass of curls?
- Charlotte Bronte, Jane Eyre
A non-conformance is a departure from an agreed arrangement between an approval authority (or orthodox position) and an individual or group performing work. In the context of quality management an "arrangement" is commonly an agreement to conform to:
- A work product standard Example: The format and content of a Software Requirements Specification does not comply with the designated SRS Document Standard
- A predefined procedure Example: The requirements elicitation activities on project X did not follow the steps required by the Joint Application Design Procedure.
- A work product specification Example: The System Architecture Specification does not address all the customer requirements provided in the System Requirements Specification.
- A documented plan Example: The activities described in the Project Plan are not being carried out.
- An international standard for best practice Example: Company XYZ does not have a documented procedure for control of quality management system documentation as required by ISO 9001 - Quality Management Systems Requirements1 clause 4.2.3 Control of documents.
|
Typical Conformance Requirements for a Work Product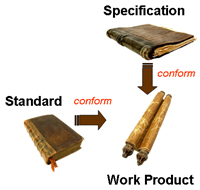 |
The Significance of a Non-conformance
The existence of a nonconformance means that some aspect of an organisation's standard operating procedures are not being followed.
How are Non-conformances Identified?
Non-conformances are typically identified in the context of a quality management system audit. The auditor documents the non-conformance in a Non-conformance Report which is subsequently issued to a management representative who directs corrective action.
Non-conformance Severity
The relative urgency of corrective action on a non-conformance is a function of its severity.
| Severity |
Description |
| Major |
Total or significant absence of objective evidence of conformance with designated criteria.
Case study:
There is no documented evidence of design review as required by ISO 9001 clause 7.3.4 Design and development review |
| Minor |
Applies where minor non-conformances are observed. For example, the inadequate implementation of a procedure.
Case study:
The design review was conducted and a Design Review Report issued. There is evidence that the design was updated but the Review Authority did not annotate the Design Review Report as required by the Design Review Procedure. |
| Trivial |
Applies where a non-conformance is insignificant or an isolated lapse of operating discipline. It can be recorded as an observation.
Case study:
Of the 300 design documents generated, one was not formally signed off by the project manager. |
Member Comments |
48 Comments |
43 member ratings |
|
✭ ✭ ✭ ✭ ✩
|
|
|

